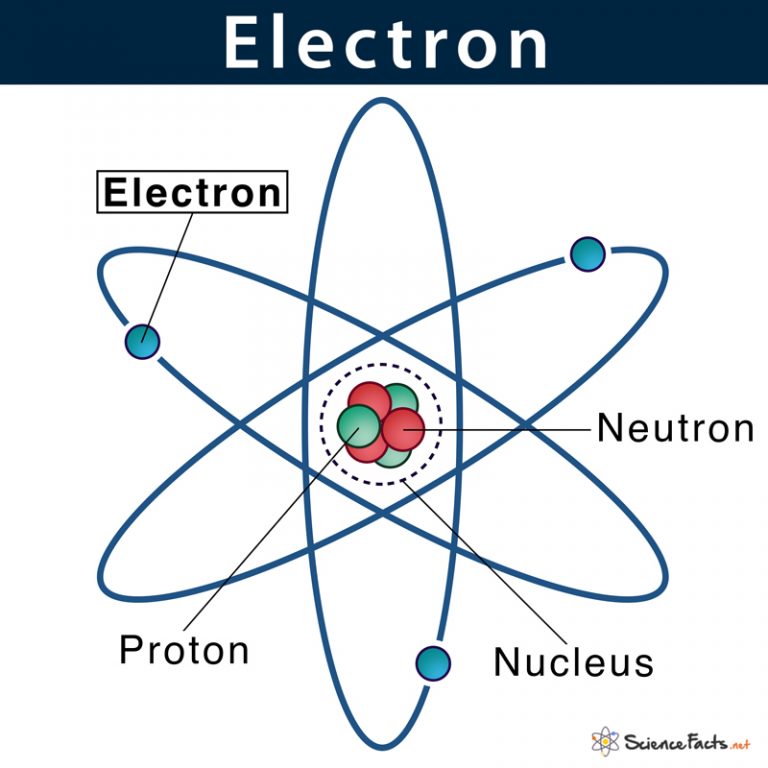
They contain particles that are positively charged and travel towards another cathode in a gas discharge tube. Read More: Difference Between Element and CompoundĪns: The positively charged radiations that can pass through perforated cathode are known as canal rays. The mass number of an atom is the number of nucleons in the nucleus.The atomic number is the number of protons in the nucleus of an atom.Rutherford’s alpha-particle scattering experiment led to the discovery of the nucleus in an atom.Quarks and leptons are significantly lower particles in an atom even though atoms are the smallest unit of an atom.Cesium is roughly multiple times greater than helium which is the smallest atom. The typical atom is around one-tenth of a billionth of a meter over which makes them exceptionally tiny.It was called Technetium and had 43 protons. The first atom was discovered by Emilio Segrè in 1937 in Italy.Neutrons and protons are held together by atomic powers that are strong and weak. The parts of an atom are held together by three powers.Usually, another atom is made by separating the tiny atoms from this rot. These new atoms or components rot into tiny atoms very fast.If the number of protons is added to the nucleus of an atom it helps in the making of new atoms. A huge percentage of them usually happen and the rest are made in labs. There are more than 100 different types of atoms.An electron’s mass is considered to be negligible and its charge is minus one. The mass of a proton is taken as one unit and its charge is taken as plus one. Protons are generally represented as p and electrons are represented as e. Its mass 2000 times that of the electron approximately. This subatomic particle had a charge which was equal in magnitude but did not share the sign the electron had. These rays were radiations that were positively charged which led to the eventual discovery of another subatomic particle. Before the electron was identified, the presence of new radiations in a gas discharge was discharged by E. By 1900, it was known that an atom was a particle that was indivisible but contained at least one subatomic particle, which is the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
